The role of sphingolipids in health and disease
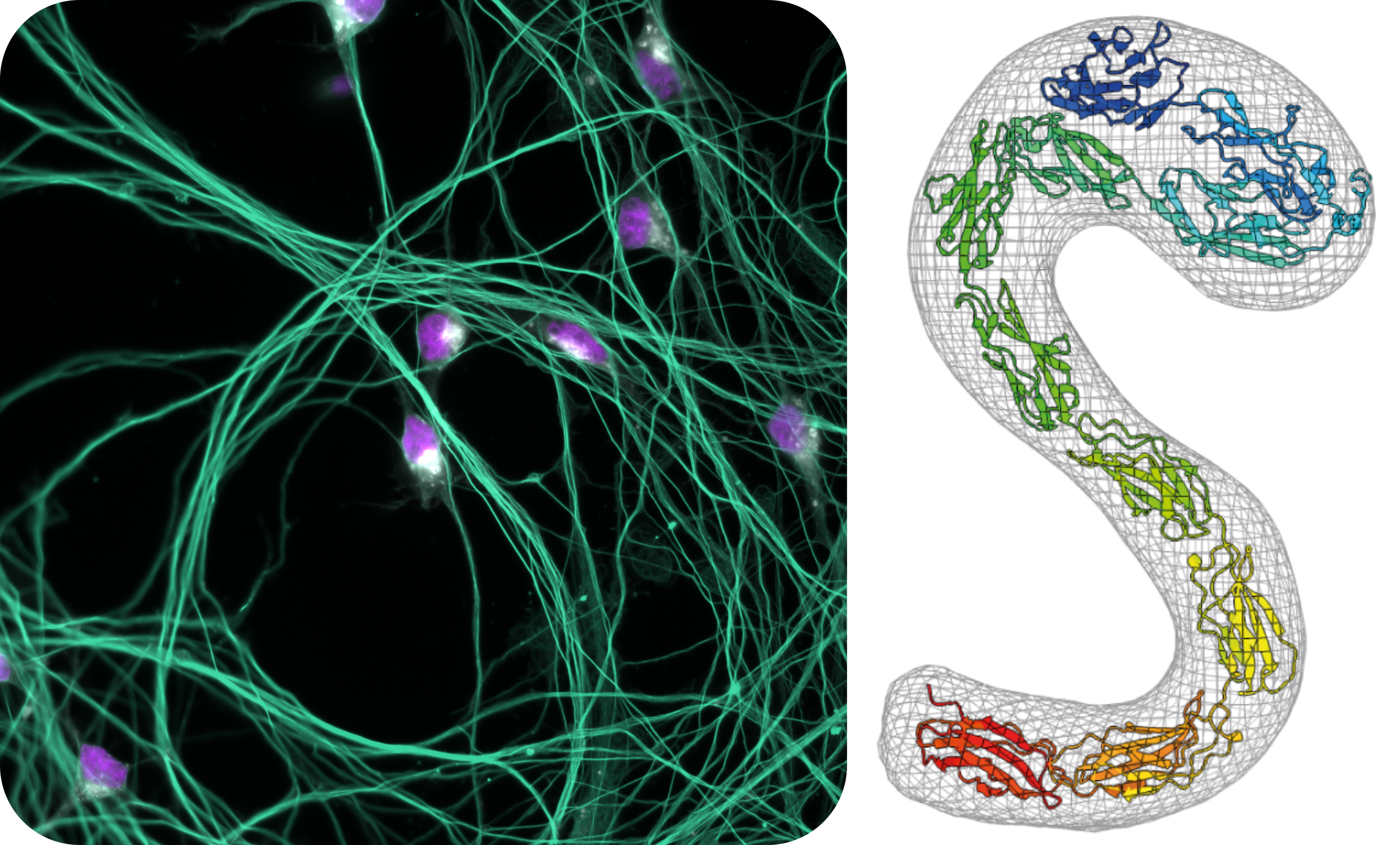
The cell surface is decorated with a diverse array of proteins and lipids that play essential roles in cell contact and signalling. An important class of lipids enriched at the cell surface are glycosphingolipids (GSLs). Imbalances in GSL levels underlie a range of severe disorders from neurodegeneration to cancer. My lab investigates the molecular mechanisms by which altered sphingolipid metabolism results in devastating neurological disease. Our work explores similarities between rare genetic neuropathologies and common neurodegenerative diseases, laying the scientific foundations for future therapies.
Strategic CIMR themes: Membrane Trafficking, Rare Genetic Disease, Neurological Disease
Funding: Wellcome Trust
Research Group Members: Henry Barrow, Ella Bishop, Zhuang (George) Han, Shannon McKie, Holly Monkhouse, Alex Nicholson, Jack Welland